Best Biomedical Devices Suppliers
KuartisMED Medical Research
🇹🇷 Turkey
201211-50 peopleManufacturerHigh-tech enterprise
Key customer
ANADOLU SIGORTA
Key market
Asia
Description
KuartisMED Medical Research is a specialized biomedical manufacturer based in Ankara, Turkey, dedicated to developing innovative medical devices for neonatal care. Founded in 2012, the company operates a manufacturing facility and focuses on the research, design, and production of smart, non-invasive diagnostic tools and AI-supported platforms specifically for preterm infants and newborns. With a team of approximately 11 to 50 professionals, KuartisMED serves as an OEM provider and has secured significant backing from international programs such as the European Union’s Horizon 2020.
The company's product portfolio is centered on enhancing clinical decision-making and infant well-being. Key technologies include NeoSAFE, a non-invasive diagnostic tool that utilizes unique sensors and artificial intelligence algorithms to objectively assess feeding maturation and respiratory-swallowing coordination in premature infants. Additionally, the company offers Nimie, an AI-supported neonatal monitoring and decision-support platform designed to automate clinical tracking for safer discharge decisions, and stetsafe, a solution focused on medical equipment hygiene.
KuartisMED maintains high quality standards, holding CE certification and the BME Seal of Approval for its electromedical equipment. By integrating medical electronics with advanced R&D, the company provides hospital-grade solutions that address critical gaps in neonatal diagnostics and pediatric healthcare.
Shenzhen Hochuen Technologies Co., Ltd.
🇨🇳 China
20042001-5000 peopleManufacturer/Trading CompanyShipments
52
Key customer
EURO CB PHILS
Key market
North America
Review
5

High Quality Medical Device Contract Manufacturing Product Development Service Custom Two Shot Injection Molding Medical$0.99

Medical Device CDMO Manufacturing Plastic Products Test Kit ABS PC PP Injection Molding for IVD and POCT Test Cartridge$0.02

Custom Silicone Rubber Pad Graphic Overlay Adhesive Keyboard Embossed Button Sticker Membrane Switch Keypad for Medical Device$0.02

ISO13485 Cleanroom Die Cut Insulation Pad Adhesive Tapes Two Side Polyurethane Polyomide 2mm Thick Foam Tape for Medical Device$0.2

ISO13485 Medical Device Cleanroom Manufacturing Factory Custom Thickness OEM Colored Double Sided Coated Adhesive Foam Tape$0.1

Custom Self-Adhesive Industrial PET Biochip Control Panel Sticker Decals Medical Device Labels Processing Service Cutting$0.2

Customization Wholesale Strong Adhesion Acrylic Clear Medical Tape Die Cut Foam Tape for Medical Device$0.05

One Stop OEM Manufacturer PP PC PS PA ABS Acrylic Molding Parts Injection Mold Making Plastic Molds Design Custom Medical Moulds$0.02

Custom Pre-cut Circle Square Shape Acrylic Adhesive Tape Waterproof Foam Tape Double Sided Adhesive Die Cutting for Medical$0.06

Medical Parts Moulding Factory Plastic Parts Machining Components Diffusion Bonding Assembly Microfluidic Chip for IVD Products$0.05

High Quality Removable Adhesive Tape Sticker Customized Fingerboard Medical Self Adhesive Foam Insulation Tape for Automotive$0.02

Product Seal Label Stamp Machined Silk Screen Printing Membrane Nameplate Custom Waterproof Embossed Sticker$0.05
Shanghai Beion Medical Technology Co., Ltd.
🇨🇳 China
2014101-200 peopleManufacturer/Trading Company/Distributor/Service CompanyShipments
1
Key customer
ELDON JAMES
Key market
North America
Description
Shanghai Beion Medical Technology Co., Ltd. is a high-tech manufacturer and service provider headquartered in Shanghai, China, specializing in in-vitro diagnostic (IVD) products and micro-fluid control systems. Established in 2014, the company operates under two primary brands: BEIONMED and BEIONFLUID. The company maintains a workforce of 101 to 200 employees and is certified under ISO 9001, ISO 13485, and ISO 14001 quality management systems. BEIONMED focuses on microscopic medical imaging and AI-driven diagnostic solutions, including sperm quality analyzers, chromosome scanning analysis systems, bone marrow blood cell image products, and automatic urine sediment analysis software. These solutions are widely utilized by over 1,500 customers, including prestigious institutions such as Peking Union Medical College Hospital and West China Hospital. Under the BEIONFLUID brand, the company serves as a microfluidic technology expert, providing comprehensive fluid system solutions for precision analysis instruments. Their extensive product portfolio includes solenoid pinch valves, isolation valves, diaphragm pumps, syringe pumps, and various high-performance connectors and PTFE tubing. These components are essential for applications in analytical chemistry, environmental monitoring, and bioengineering. With a strong emphasis on R&D, the company holds over 120 patents and offers OEM and ODM services to global partners. Shanghai Beion Medical Technology Co., Ltd. actively participates in international trade, exporting its high-quality medical and fluid control products to markets across North America, Europe, and Southeast Asia.

Thermoplastic Elastic Tube BFT045 Biomedical Standard Tube without Animal Additives$20.8

Thread turn barb connector hose connector quick coupler 1/4 "-28unf & 10-32unf & 10-32taper & 1/16" NPT & M6$0.45

Hose barb quick pagoda thread adapter hose fitting socket right angle thread adapter$0.89

Thread turn barb connector hose connector quick coupler 1/4 "NPSM$0.62

Hose Connector Screw Adapter Hose Pipe Fittings Pullover Quick Connector Right Angle Flow Reducer$0.92

Straight Connector Quick Pagoda Connector Hose Adapter Reducer RoHS Biopharmaceutical Connector$0.52

Cross barb connector, Baota equal diameter hose adapter, ROHS, no animal additives$1

Thread Turn Barb Connector Hose Connector Quick Coupler 1/4 "-28UNF$0.76

Thread Turn Barb Connector Hose Connector Quick Coupler 1/8" NPT & More$0.64

Thread Turn Barb Connector Hose Connector Quick Coupler 1032 UNF & 1428 UNF$0.64

Flanging and pipe cutting tools Flange flanging pipe set$25
Shenzhen GSD Tech Co., Ltd.
🇨🇳 China
1995201-300 peopleManufacturer/Trading Company/Service CompanyShipments
133
Key customer
SKINNOVATION
Key market
North America
Review
5

Sapphire IPL Laser Hair Removal Device for Home Salon Use Professional Portable Ipl Hair Removal Affordable Epilator for Women$92

High-Power Home IPL Laser Hair Removal Skin Rejuvenation Device with Unlimited Flashes Ipl Machine at Home$88

DEESS IPL Hair Removal Device for Body Permanent Laser Epilator Professional Ipl Laser Hair Remover for Home Use$92

Painless Hair Removal Permanent Ice Cooling IPL Hair Removal Laser Machine Portable IPL Hair Removal Device for Face Body$88

Hair Removal Electronic Machine Hair Removal Laser IPL Hair Remover Permanent Hair Removal Device Home Use$92

Professional Home & Salon IPL Laser Hair Removal Machine 2023 High-Efficiency Permanent Ipl Devices Hair Removal Solution$105

DEESS Professional IPL Hair Removal Machine Ice-Cooled IPL Laser Hair Removal Device Home Use with 5-Level Intensity Adjustment$90

DEESS GP591C Ice CoolingPainless Handset Permanent Unlimited Flashes Ipl Hair Removal Laser Device Hair Removal Epilator$50

DEESS GP592 SmartBeauty IPL Laser Epilation Hair Removal Device for Home & Salon Use Laser Hair Removal Device$90

Ipl Hair Removal Device at Home Permanent Laser Hair Removal Device for Body Painless Hair Removal Machine for Women DEESS$105
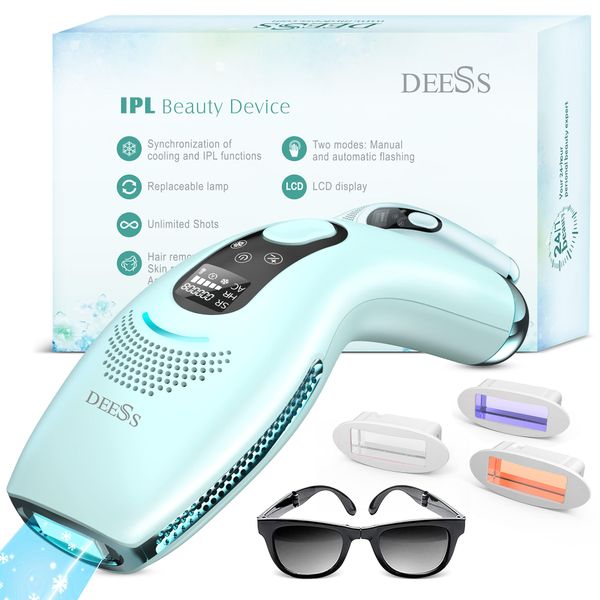
DEESS 100% Certification Permanent and Painless Home Use Portable Ipl Laser Har Removal DeviceIPL SR+HR+AC$50

DEESS Hair Removal Epilator Ice CoolingPainless Permanent Unlimited Flashes Hair Removal Laser Device Laser at Home$50
Qinhuangdao Huisianpu Medical Systems Inc.
🇨🇳 China
2011101-200 peopleManufacturer/Trading CompanyShipments
7
Key customer
WELLTEC
Key market
🇩🇪 Germany
Review
2.4

Portable Medical Grade PEMF Therapy Device Exclusive System Physical Therapy Equipment for Joint Pain Relief and Improved Sleep$249

Best Choice Professional Medical Devices Optimal Magnetic Therapy Physical Therapy Machine for Medical Treatment$900
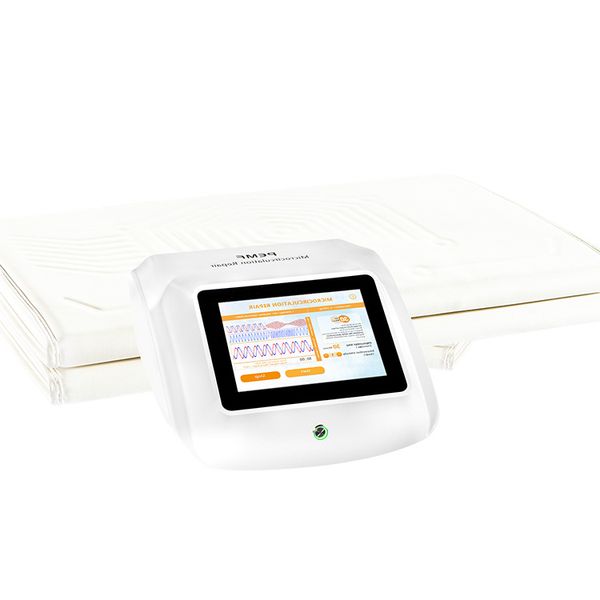
Pulse Magnetic Infrared Therapy Device for Microcirculation Repair Physical Therapy Equipment for Body Leg PMR Medical Equipment$900

No Side Effect Pemf Magnetic Therapy Device Sleep Aid for Hand Lower Limbs Arthritis Physiotherapy Physical Therapy Equipment$900

FDA-Certified Home Use PEMF Mat Certified Household Medical Device for Pain Relief Sleep & Detox Magnetic Therapy Device$450

Professional Medical Devices for Rehabilitation and Physical Therapy Quality Guaranteed for Bone Improvement on Sale$900

2025 Portable High Potential Therapy Machine for Constipation Effective Physical Therapy Equipment$900

Factory Manufacture Anti-Aging Physiotherapy Rehabilitation Equipment Imported Curing Neck Pain Physical Therapy Gear$500

Large-Capacity 90-Litre Pet Dryer Pulsed Electromagnetic Field (PEMF) Therapy Intelligent Temperature Veterinary Instrument$200

AM6200 Pet Monitor One-Button Operation Measures ECG Heart Rate Non-Invasive Blood Pressure Blood Oxygen Saturation Pulse Rate$260

Hot Sale Portable Home Physiotherapy Equipment Top Physical Therapy Gear for Effective Joint Pain Relief$249

Home Painless Veterinary Instrument Magnetic Therapy Equipment for Pets Immunity & Gastrointestinal Conditioning$500
Lucid Labs
🇺🇸 United States
200411-50 peopleManufacturer/Service CompanyKey customer
3M
Description
Lucid Labs is a US-based manufacturer and service company located in Rogers, Minnesota, specializing in medical device product development and contract manufacturing. Established in 2004, the company employs 11-50 people and is certified with ISO 13485:2016, demonstrating its commitment to quality in the medical sector. Lucid Labs offers comprehensive services from concept to FDA submission, including agile product design and development for complex medical device implantables, delivery systems, and external rechargeable products. Their manufacturing capabilities include 3, 5, 7, and 9-axis precision CNC machining, milling, turning, wire EDM, laser cutting, water jet cutting, injection molding, and additive manufacturing (3D printing) for both metal and plastic materials. They also provide PCB design, prototyping, and assembly services. Lucid Labs serves a diverse client base, from medical device startups to Fortune 500 companies like Medtronic, 3M, Abbott, and Johnson & Johnson, accelerating product development cycles with rapid in-house capabilities and world-class responsiveness.

bsci

infraredsolutions

COMPREHENSIVE, DEEP EXPERTISE

COMPREHENSIVE, DEEP EXPERTISE

AGILE PRODUCT DESIGN & DEVELOPMENT

Corventis_logo

r4vascular

DEVELOPMENT AND PROTOTYPING TO SHORT-RUN PRODUCTION AND MANUFACTURING

johnson-johnson-auris-square_blue

DENTAL SCANNER DESIGN

heartware

NuXcel-Logo_square_blue
Humaneotec Health Industry (Guangdong) Co., Ltd.
🇨🇳 China
200811-50 peopleManufacturer/Trading CompanyShipments
5
Key customer
AGARWAL ENTERPRISE
Key market
Asia
Review
5
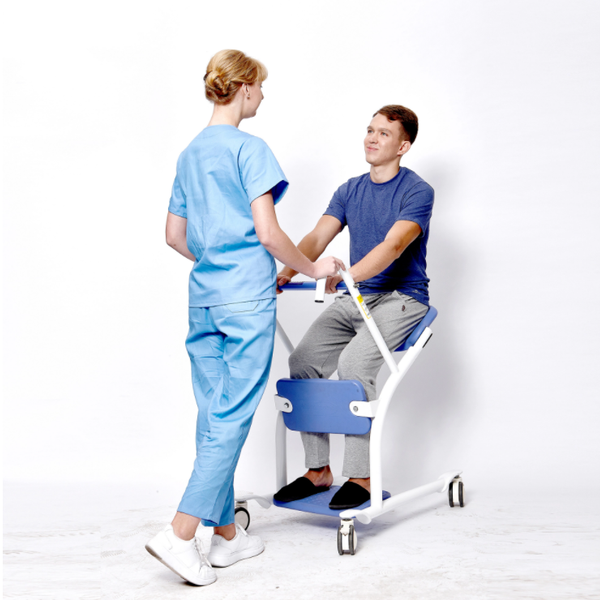
Rehabilitation Device Portable Walker Chair for Adults$428

Humaneotec Physiotherapy Rehabilitation Equipment Stroke Elderly Transfer Lift Devices Standing Transfer Aid$500

Physiotherapy Rehabilitation Sport Equipment for Stroke Exercise Device Recumbent Cross Trainer$3095

Electric Shock Stimulation Machine Pain Relief Device Recovery of Lower Limb Dysfunction and Promotes Leg Rehabilitation$1110

Multi-function Therapy Device Red Light Pain Relief Infrared Lamp Light with Stand$35.8

Medical Rehabilitation Physiotherapy Exercise Device Recumbent Cross Trainer for Cardiopulmonary Rehabilitation$3200

Rehabilitation Parallel Bars Height Adjustable Physical Therapy Equipment Walking Training Device for Adults and Kids$260

Rehabilitation Equipment Foldable Patient Lift Device with Walking Sling for Patient Transfer$1499
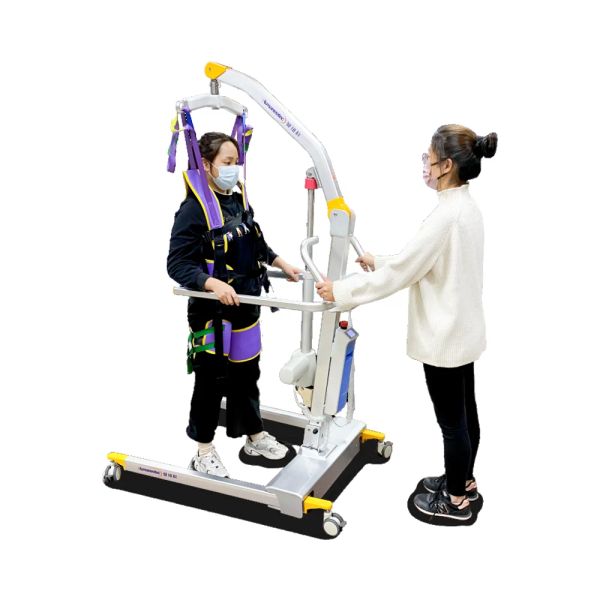
Ergonomic Design Patient Transfer Gait Training Exercise Device for Walking Rehabilitation$1199
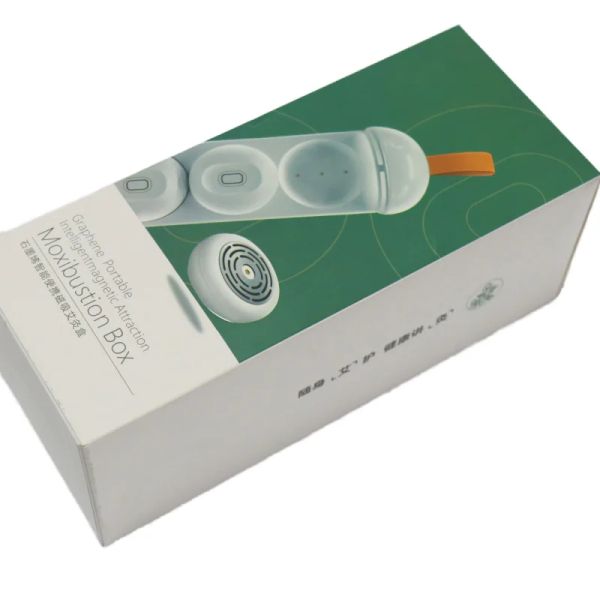
Traditional Chinese Medicine Rechargeable Smokeless Moxibustion Device With Graphene Heating Massager Product$22.5
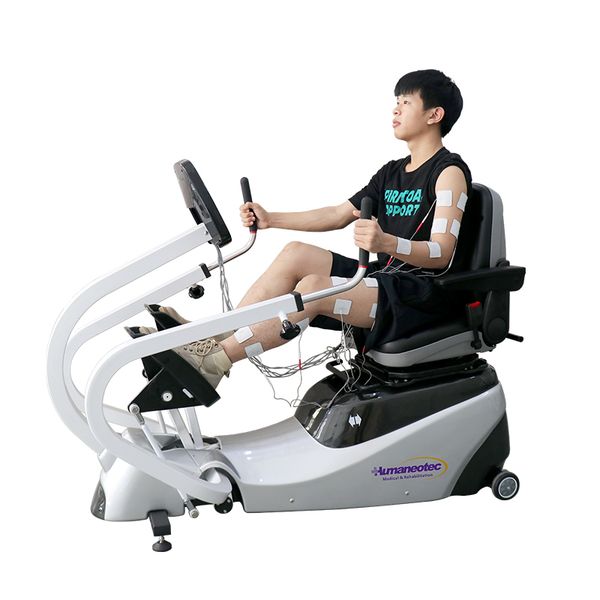
Medical Physiotherapy Recumbent Cross Trainer Sports Bike Exercise Rehabilitation Device for Rehabilitation Equipment$3200

Rehabilitation Equipment for Stroke Exercise Device Recumbent Cross Trainer with 32 Level Resistance$3095
Omnica Corporation
🇺🇸 United States
198411-50 peopleManufacturer/Service CompanyKey customer
ABBOTT MOLECULAR
Key market
North America
Description
Omnica Corporation, now a part of StarFish Medical, is a premier medical device design and engineering firm headquartered in Irvine, California. Founded in 1984, the company operates from a 25,000-square-foot facility equipped with advanced in-house machining, model making, and rapid prototyping capabilities. As a contract manufacturer and service provider, Omnica specializes in the end-to-end development of complex medical technologies, including diagnostic products, surgical instruments, point-of-care devices, and monitoring equipment. Their multidisciplinary team of experts provides comprehensive services across industrial design, mechanical and electronics engineering, software development, and regulatory affairs consulting. Holding ISO 13485 and FDA certifications, the company supports a diverse clientele ranging from early-stage startups to global MedTech leaders like Medtronic and Abbott. By integrating with StarFish Medical, Omnica leverages expanded North American resources to deliver specialized R&D, feasibility studies, and seamless transitions to manufacturing for high-technology medical products.

Medical Device Design & Development

Medical Device Manufacturing Support

Surgical & therapeutic systems

Surgical & therapeutic systems
Kernel Medical Equipment Co., Ltd
🇨🇳 China
1997501-1000 peopleManufacturer/Trading CompanyShipments
463
Key customer
DOLPHIN
Key market
🇦🇫 Afghanistan
Review
4.8
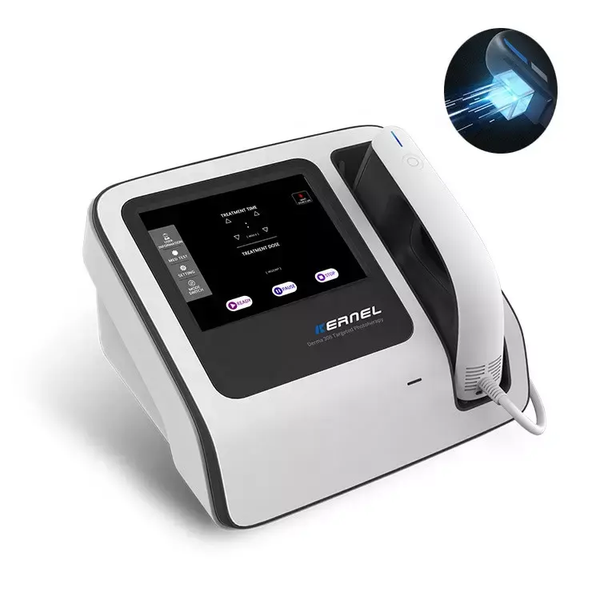
Kernel Factory Precise Treatment 308nm Excimer Device High Power Best Machines Supplies Treatment of Vitiligo Eczema$1800

Dermatoscopio Para Detectar Verrugas Kernel Factory Wood Lamp Light Skin UV Analyzer Facial Care Magnify Skin Testing Device$99

Medical Equipment 308nm Excimer Laser Light Phototherapy Device Physical Therapy Vitiligo Psoriasis Treatment CN-308C$1600

KN 4006B Household Medical Device New Trend Atopic Eczema Vitiligo Treatment Ultraviolet Narrow Band Hand-held UV Lamp for$220
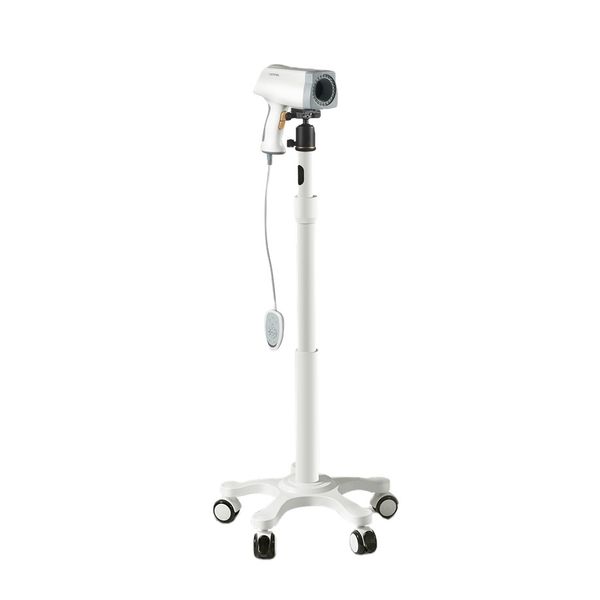
Kernel KN2200 Colposcope for Gynecology Colposcope Price Medical Colposcope Device$1300

Hair Loss Treatment Device Kernel KN-8000A 424 Pcs Lasers Lllt Hair Growth Machine for Alopecia Areata Treatment$3100

KN-8000B Red Light Therapy Hair Regrowth Hat 650nm LLLT Therapy Laser Helmet Cap for Hair Growth Laser Hair Growth Device$499

650nm Cold Laser Therapy Machine Medical Laser Hair Growth Device$3100

Kernel Uvb 311nm Uvb Phototherapy Light Therapy Ultraviolet Household Devices$180

For Kernel KN-5000F Handheld 308nm Excimer Laser UV Phototherapy Medical Device for Vitiligo Psoriasis Eczema Treatment$1000

Kernelmed CN-308P6 308 nm LED Light Devices for Cost-effective Alternative to In-office Treatments with 60mW/cm2$400

Kernel 2025 KN-4003BL2 Home Vitiligo Phototherapy Device Portable 311nm UVB Light Therapy for Psoriasis & Vitiligo Treatment$180
Shanghai Sunbright Industrial Co., Ltd.
🇨🇳 China
1998501-1000 peopleManufacturer/Trading CompanyShipments
47
Key customer
YOGI
Key market
South America
Review
4.8

Rectal Linear USB Probe Medical Devices Suppliers Color Doppler Veterinary Use$299

15 Inch Remote Patient Monitoring Devices Hospital Digital Vet Patient Monitor Machine$379
Medical Devices Suppliers Rectal Convex BW Color Doppler Probe Veterinary Equipment$199

15 Inch PET Patient Monitoring Devices Hospital Digital Vet ICU Patient Monitor Machine$99

Veterinary Middle Pregnancy Rectal Convex USB Color Doppler Probe Medical Devices$199

Hot Veterinary X-ray Vet Photo Camera Machine DR for Medical Imaging Center for Pet Diagnosis X-ray Device$4899

Horse Cow Middle Pregnancy Rectal Convex Color Doppler Probe Veterinary Scanner$199

USB Rectal Linear Color Doppler Probe Animals Early Pregnancy Probe Farm Use$299

Rectal Linear Color Doppler Probe Veterinary Farm Use Ultrasound Probe$299
SUN-807F Portable Veterinary USG Handheld Ultrasound Machine Ultrasound for Sector Animal Clinic Pet$100

Good Quality Cheap Hot Selling 5KW Nice Price 10.4 Inch Touch Screen Portable X-ray Machine$4899

High Resolution Veterinary ECG Instrument Portable 3 Channel 12 Lead Electrocardiograph ECG$199