Best Medical Device Components Suppliers
Shenzhen Hochuen Technologies Co., Ltd.
🇨🇳 China
20042001-5000 peopleManufacturer/Trading CompanyShipments
52
Key customer
EURO CB PHILS
Key market
North America
Review
5

Customization Wholesale Strong Adhesion Acrylic Clear Medical Tape Die Cut Foam Tape for Medical Device$0.05

High Quality Medical Device Contract Manufacturing Product Development Service Custom Two Shot Injection Molding Medical$0.99

ISO13485 Medical Device Cleanroom Manufacturing Factory Custom Thickness OEM Colored Double Sided Coated Adhesive Foam Tape$0.1

Custom Self-Adhesive Industrial PET Biochip Control Panel Sticker Decals Medical Device Labels Processing Service Cutting$0.2

Custom Silicone Rubber Pad Graphic Overlay Adhesive Keyboard Embossed Button Sticker Membrane Switch Keypad for Medical Device$0.02

ISO13485 Cleanroom Die Cut Insulation Pad Adhesive Tapes Two Side Polyurethane Polyomide 2mm Thick Foam Tape for Medical Device$0.2

Medical Device CDMO Manufacturing Plastic Products Test Kit ABS PC PP Injection Molding for IVD and POCT Test Cartridge$0.02

Medical Parts Moulding Factory Plastic Parts Machining Components Diffusion Bonding Assembly Microfluidic Chip for IVD Products$0.05

Tamper-Evident Anti-Counterfeiting Security Stickers High-Visibility Warning Industrial Labels Certified Medical Label$0.02

OEM Medical Product PSA Strong Adhesives 3M Custom Die Cutting Round 9448A Electricity Adhesive Masking Tape$0.05

High Quality Removable Adhesive Tape Sticker Customized Fingerboard Medical Self Adhesive Foam Insulation Tape for Automotive$0.02

Custom Die Cut Medical Products Different Shape 3M Double Adhesive Sided Foam Tape for Cushioned Foam Shockproof$0.05
Hochuen Medical Technology Co., Ltd.
🇨🇳 China
2015501-1000 peopleManufacturer/Trading CompanyShipments
19
Key customer
TASSO
Key market
South America
Description
Hochuen Medical Technology Co., Ltd. is a leading global OEM, ODM, and CDMO manufacturer specializing in medical disposables and devices for the life sciences, IVD, and drug delivery sectors. Headquartered in Shenzhen, China, with a significant international presence including offices in Los Angeles and Singapore, the company operates advanced manufacturing facilities in Malaysia and China. With a workforce of 501 to 1,000 employees, Hochuen provides end-to-end turnkey services ranging from R&D and rapid prototyping to high-volume production. Their extensive technical capabilities include precision injection molding, microfluidic chip fabrication, die-cutting, CNC machining, and specialized bonding techniques such as laser and ultrasonic welding. The company is well-equipped for complex medical manufacturing, featuring Class 10K-100K cleanrooms and GMP-compliant facilities. Their product portfolio encompasses biomedical consumables, medical wearables, biosensors, and reagent packaging. Hochuen Medical is FDA registered and holds ISO 13485, ISO 14001, and GMP certifications, serving over 250 global clients including major industry leaders in diagnostics and medical technology.

Custom Die Cutting Self Adhesive EVA Foam Gasket for Medical Use$0.02
Shanghai Haotianjia Technology Co., Ltd.
🇨🇳 China
2021101-200 peopleManufacturer/Trading CompanyShipments
9
Key customer
QUICKDENT
Key market
🇮🇳 India
Description
Shanghai Haotianjia Technology Co., Ltd. is a professional manufacturer and trading company specializing in medical sterilization solutions and precision plastic engineering. Based in Shanghai with a significant production facility in Dongyang, Zhejiang, the company operates a 3,000-square-meter mold injection workshop and a 2,000-square-meter assembly workshop. Established with a focus on innovation, the firm employs between 101 and 200 personnel and maintains an annual output value between US$10 million and US$50 million.
The company specializes in the production of medical autoclave and sterilization boxes, including dental implant kits, surgical instrument boxes, and silicone disinfection trays. Utilizing advanced polymer materials such as PSU, PPSU, and PEEK, Haotianjia Technology employs high-temperature molding processes where mold temperatures reach 200°C and injection temperatures reach 380°C. Their product portfolio is categorized into planting, repair, and orthopedic series, often incorporating specialized magnet components like NdFeB and SmCo.
As an OEM and ODM service provider, the company offers end-to-end precision plastic solutions from initial product design and mold manufacturing to secondary processing and final assembly. Their capabilities serve diverse high-precision industries, including medical stomatology, aviation lighting, consumer electronics, and home appliances. Shanghai Haotianjia Technology Co., Ltd. holds CE certification for its products and maintains an active international trade presence, exporting medical kits and surgical instrument containers to global markets, including partners in India.

Planting Drill Bit Group Immediate Planting Tool Kit Joint Diagnosis and Treatment Disinfection Box on Sale$49

Digital Management System Disinfection Box Electric Planting Machine Accessories Screw Fixing Tool on Sale$49
Vonco Products, LLC
🇺🇸 United States
195551-100 peopleManufacturer/Wholesaler/DistributorShipments
5
Key customer
ZETA BIOSYSTEM
Key market
🇺🇸 United States
Description
Vonco Products, LLC is a premier US-based contract manufacturer specializing in custom flexible packaging and medical device solutions. Founded in 1955 and headquartered in Trevor, Wisconsin, the company operates a state-of-the-art facility that includes cleanroom manufacturing and advanced assembly capabilities. Vonco serves as a turnkey partner for leading global brands and healthcare providers, offering a comprehensive "Roadmap to Commercialization" that spans from initial concept and design to scalable prototyping, sterilization management, and final distribution.
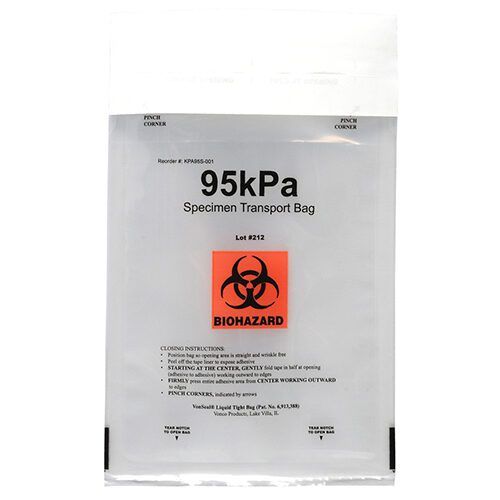
Medical Device Covers

Medical Device Covers

Blood Storage Device

Blood Storage Device

Medical Bladder with Multiple Mandrel Welds

Medical Bladder with Multiple Mandrel Welds

Media Reservoir Bag

Media Reservoir Bag

Elution Kit Component for Air Sampling Analysis

Zipper Pouches

Spouted Stand-Up Pouches

Evidence Property Bags, VonSeal®
CHI FENG CO., LTD.
🇹🇼 Taiwan
198851-100 peopleManufacturer/Trading CompanyShipments
1143
Key customer
CAMBODIA SUMMIT WOODWARE
Key market
North America
Review
5

Clear Neutral Needleless Plastic Medical Connector for Manufacturer's Medical Devices$0.3

Air Medical Transport Dialysis Procedures IV Line Connector Medical Plastic Product$0.3

High Quality Disposable Medical Transfusion Joint Needleless Connector ISO13485 Certified Plastic Medical Consumables$0.21

Multiple Color PP Medical Tube Clamp$0.04

Medical Grade Plastic Multi-Dose Vial Adapter$0.22

Nurse's IV Line Connector Medical Plastic Product$0.3

Medical Plastic Catheter Connector for Chemotherapy Clinics$0.21

Medical Plastic Needleless Connectors for Chemotherapy Centers$0.3

Needleless Medical Plastic Connector for Surgical Centers$0.21

Blue Male Luer Connector Medical Plastic Product Closed$0.9

Transparent Medical Plastic IV Infusion Needle Free Connector$0.29

Medical Grade Needle-Free IV Access for Hospitals$0.33
Wuxi Gelanten Technology Co., Ltd.
🇨🇳 China
202411-50 peopleManufacturer/Service CompanyShipments
2
Key customer
KNITTING ZONES
Key market
North America
Description
Wuxi Gelanten Technology Co., Ltd. is a professional manufacturer and service provider based in Wuxi, China, specializing in precision component machining and tailored technical solutions. Leveraging over 20 years of industry expertise, the company operates as a contract manufacturer offering comprehensive OEM and ODM services. Their core capabilities include CNC milling, turning, grinding, wire EDM, metal powder injection molding (MIM), sheet metal fabrication, and precision casting. Gelanten serves a diverse range of high-precision industries, including defense, automotive, medical devices, robotics, aerospace, and consumer electronics.
The company is recognized for its production of mission-critical components such as transmission shafts, surgical instrument joints, titanium alloy medical parts, and custom auto accessories. Their facility is equipped with advanced machinery, including CNC machining centers, torsion spring machines, and specialized welding equipment. To ensure rigorous quality standards, Gelanten utilizes high-precision inspection tools such as coordinate measuring machines (CMM), optical projectors, and altimeters, maintaining compliance with FDA 510(k) testing and DIN 876-1 standards.
With a workforce of 11 to 50 employees, the company manages the entire production lifecycle from initial concept and toolmaking to assembly and surface treatments like passivation, anodizing, and heat treatment. Gelanten maintains a global client base across North America, Europe, and Asia, providing cost-effective, high-efficiency manufacturing solutions for complex industrial components.

Professional High-Precision Die Casting Services for Brass Zinc Stainless Steel Iron Aluminum Wheels & Components$0.1

High-Demand Precision CNC Processing for Custom Mold Forming of Aluminum Components Custom Casting Services$0.1

Manufacturer of Custom Plastic Injection Molds Precision Mold Service for Plastic Products$3.2

High Precision Custom Rapid Prototype Cnc Milling Lathing Metal Parts CNC Turning Machining Services$0.1

Factory Outlet Brass & Stainless Steel Micro Machining Customized Logo Injection Molds Tooling Fixture$3.2

High Precision Custom Metal Manufacturing for Industrial Optical Lens Tooling Industrial Optical Lens Moulds$3.2

Custom High Precision 4-5 Axis CNC Machining Services for Titanium Stainless Steel Brass Aluminum-Milling & Turning Parts$0.1

High Precision Plastic Injection Mold and Molding Service for Customized Plastic Housing Parts Supplier$3.2

Custom Manufacturing CNC Turning Services Precision Lathe Parts Aluminum/Steel/Stainless Steel Fast Prototype CNC Service$0.1
Shanghai Yingzhou Metal Products Co., Ltd.
🇨🇳 China
201511-50 peopleManufacturer/Trading CompanyShipments
34
Key customer
LIBRA MARINE
Key market
Oceania
Description
Shanghai Yingzhou Metal Products Co., Ltd. is a professional manufacturer and trading company established in 2015 and based in Shanghai, China. Operating a factory facility of up to 3,000 square meters, the company specializes in providing one-stop solutions for custom sheet metal fabrication and precision assemblies. Their core manufacturing capabilities include plate and tube laser cutting, CNC machining, stamping, punching, bending, welding, and powder coating.
The company serves a diverse range of industries, with a significant focus on medical device parts, mechanical equipment, ship fittings, and automotive spare parts. Notably, they are a recognized supplier for Shanghai SIEMENS Medical Device Co., Ltd. Their product portfolio features stainless steel frames, electrical cabinets, aluminum enclosures, metal brackets, and conveyor components. Beyond fabrication, they offer comprehensive OEM, ODM, and design services, supported by a dedicated technical team and over 10 production lines.
Shanghai Yingzhou Metal Products Co., Ltd. maintains a strong commitment to quality and international standards, holding ISO 9001 and CE certifications. With an annual revenue between US$5 million and US$50 million and a workforce of approximately 11 to 50 employees, the company is an active exporter with its own export license. They have established a global trade presence, particularly in the United States, serving clients such as Libra Marine, LLC and Strong Industries Inc.
Dongguan Huayingxin Technology Co., Ltd.
🇨🇳 China
201911-50 peopleTrading CompanyShipments
67
Key customer
VINA CNS
Key market
North America
Description
Dongguan Huayingxin Technology Co., Ltd. is a professional trading company based in Dongguan, China, specializing in high-precision metal components and assembly services. Established in 2019, the company has quickly positioned itself as a versatile supplier of Metal Injection Molding (MIM), CNC machining, and die-casting solutions. Their extensive product portfolio includes powder metallurgy parts, 5-axis machining components, and specialized hardware for the medical, consumer electronics, and automotive industries.
The company offers a wide range of custom-engineered products, such as mobile phone hinge components, TWS earphone shells, watch accessories, and power tool parts. They are particularly noted for their expertise in manufacturing complex items like steel bevel gears, aluminum housing products, and zinc alloy die-cast parts. Their technical capabilities extend to processing various materials, including stainless steel, brass, and anodized aluminum.
Operating with a workforce of 11 to 50 employees, Dongguan Huayingxin Technology Co., Ltd. maintains a strong commitment to quality and international standards, holding ISO 9001, ISO 14001, and ISO 45001 certifications. As a verified supplier, they emphasize high-quality production, integrity, and a 100% on-time delivery rate. The company is actively involved in international trade, with a significant export presence in markets such as Vietnam, where they supply specialized components like steel auxiliary arm joints and metal spacers to global manufacturing partners.

Recommended 304 Stainless Steel Machinery Hardware for Powder Processing Medical Metallurgy Metal Metallurgy Machinery Parts$0.77

High Quality Stainless Steel Powder Injection Medical Hardware Parts Accessory Equipment for Metal & Metallurgy Machinery$0.77

OEM ODM Custom Stainless Steel Medical Surgical Instrument Feeder Parts MIM Powder Metallurgy Sintered Metal Machining Services$0.8

OEM Professional Factory's Iron-Based Ferrules Chinese PM Factory Price Metal Component Broaching Sintering Powder Metallurgy$0.1

Direct Factory Sale New TC4 Titanium Alloy Parts MIM Powder Metallurgy Hardware Components Chrome Metal Metallurgy Machinery$0.77

Customized 5-Axis CNC Turning Service Metal Stainless Steel Prototype CNC Machined Parts$0.79

MIM Powder Metallurgy Stainless Steel Metal Injection Molding Parts Pink Silver Gradient Color Buckle Metal Machining Services$0.1

Customized Bronze Brass Bushing | Custom Self-Lubricating Flanged Sleeve For Industry Use$0.1

Wholesale Precision Casting Services CNC Machining for Stainless Steel & Copper Brass Lost Wax Casting Broaching Included$0.1

Custom-Made CNC Machining Accessories Stainless Steel Bearings for Bags Metal & Metallurgy Machinery Broaching Type$0.1

Custom High Quality Precision Zinc Alloy Automatic Die Casting Service for Heat Sink Housing$0.1

Professional Factory China High Quality MIM Service for Powder Metallurgy Stainless Steel Metal Injection Molding Services$0.1
Shenzhen Honvision Precision Technology Co., Ltd.
🇨🇳 China
2001101-200 peopleManufacturer/Trading CompanyShipments
3
Key customer
SPEROGENX BIOSCIENCES
Key market
North America
Review
5
Milling center parts , high precision parts, used for CNC machining, ISO 9001:2008 certified
Ningbo Five Continents Medical Instrument Co., Ltd.
🇨🇳 China
200451-100 peopleManufacturer/Trading CompanyShipments
26
Key customer
PFO
Key market
🇪🇪 Estonia
Review
4.9

Disposable Surgical Lavage System Irrigation Device Wound Cleaner Arthroplasty Medical Device Fast$21

Surgical Device Disposable Electrosurgical Medical Blue Esu Pencil$0.5
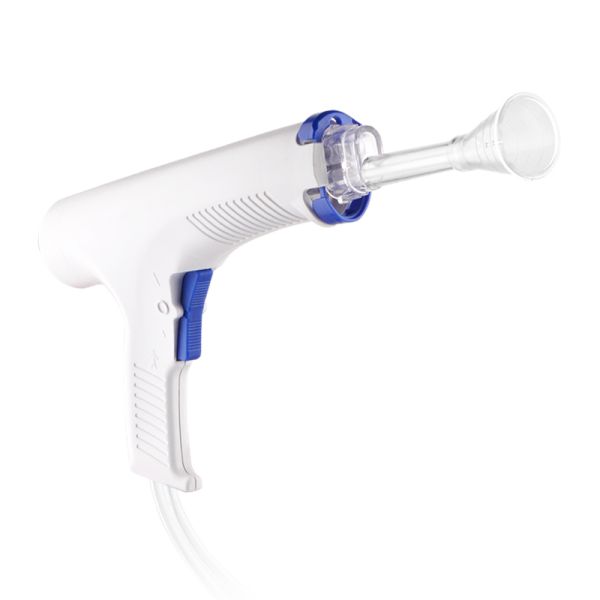
Innovative Disposable Wound Irrigator Arthroplasty Medical Device Technology$21
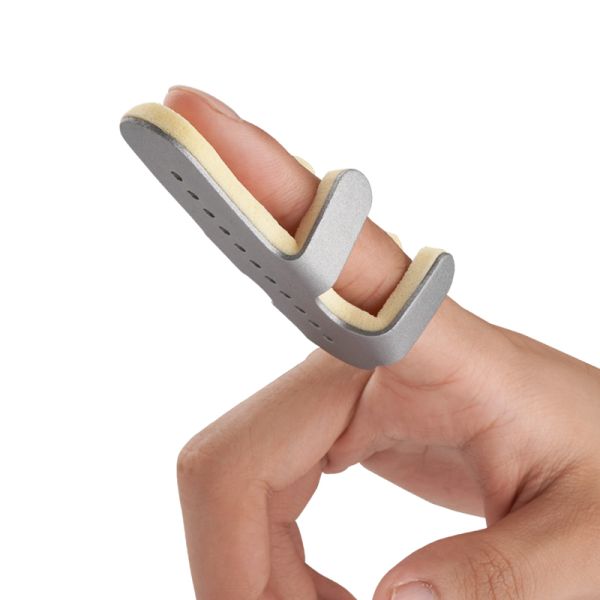
Qualified Medical Care Device Finger Splint Orthopedic Splint$0.5

Disposable Surgical Lavage C Wound Cleaner Arthroplasty Medical Device$21
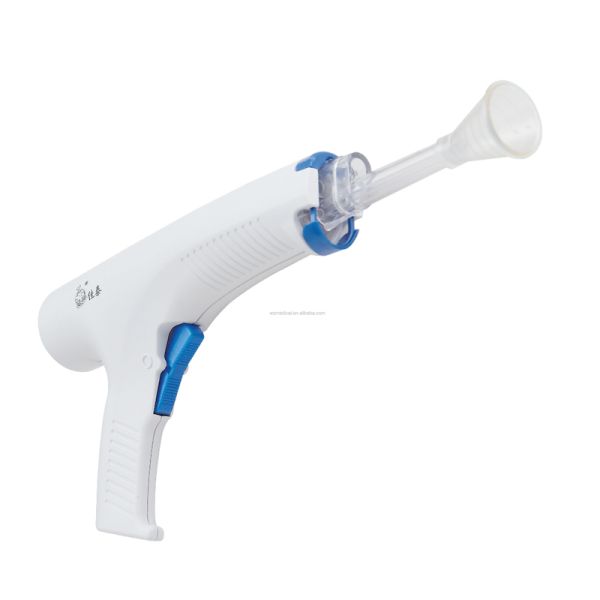
Medical Device Pulse Lavage in Arthroplasty Surgery and Truma Cases$10

Disposable Pulse Lavage System Surgical Wound Cleaner Arthroplasty Medical Device Fast$21
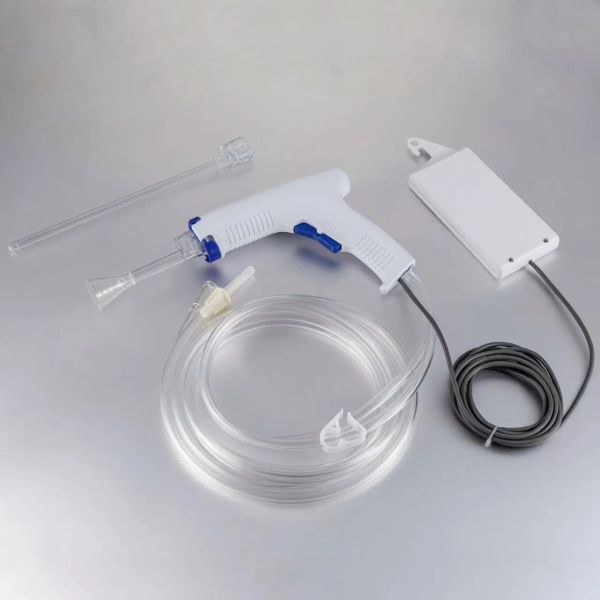
Disposable Pulse Lavage System Irrigator Surgical Wound Cleaner Arthroplasty Medical Device$21

High Frequency Medical Devices Diathermy Pencil Manual Unipolar Electrodes Electrosurgical Pencil$0.5

Disposable Surgical Lavage System Irrigator Surgical Wound Cleaner Arthroplasty Medical Device Fast$21

CE & ISO Disposable Hospital Surgical Lavage System Medical Device Wound Debridement Irrigation$18
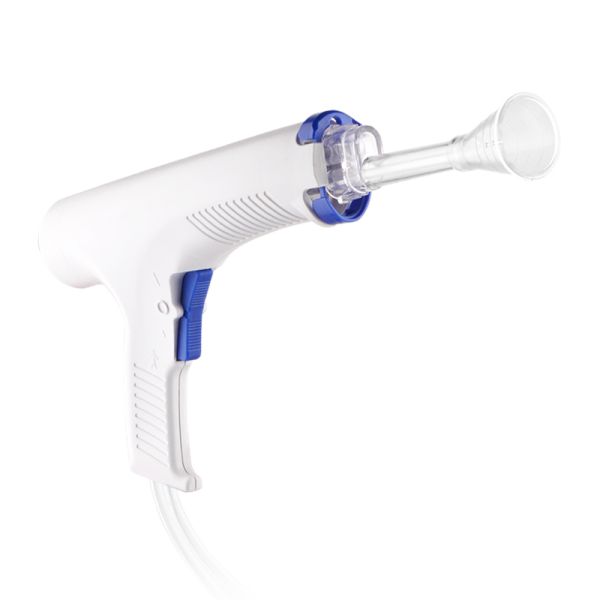
Disposable Pulse Lavage System Irrigator Surgical Wound Cleaner Arthroplasty Medical Device Fast$21